What drives chronic neck pain? Insights from human neural tissue
- Michele Curatolo

- Apr 23
- 3 min read

Chronic neck pain is very common and is a leading cause of disability. A major barrier to developing effective treatments is the very limited understanding of which biological pathways are involved in the generation and elaboration of painful signals.
A rare opportunity to study human neck pain
Pain signals from the neck travel through small bundles of nerve cells called dorsal root ganglia, or DRGs, which are located just outside the spinal cord. The DRG at the second cervical level (C2) is especially important because it carries signals not only from the neck but also from the back of the head. The DRGs from living individuals are almost never available for research. However, during certain spine surgeries, the C2 DRG is sometimes removed as part of routine care. In our study [1], we examined this tissue to better understand the changes that occur in the cells when pain becomes chronic.
What we did
We analyzed the DRG from 22 patients:
Half had recent neck pain (lasting less than 3 months)
Half had chronic neck pain (lasting 3 months or longer)
Before surgery, patients also completed surveys and sensory testing to link biological findings to pain experiences.
We used advanced tools, called RNA sequencing, that allowed us to:
Measure which genes were active in individual cells
See where those cells were located within the DRG
Compare patterns between people with acute versus chronic pain
This gave us a detailed picture of what is happening inside the C2 DRG in patients with neck pain at the single-cell level.
What we found
The C2 DRG looked partly like nerves that supply the face and head
The similarity in molecular composition helps explain why neck problems can sometimes cause headaches or pain at the back of the head.
The surprise: the changes are not in the nerves
We expected to see major changes in the nerve cells (neurons) of the DRG that transmit pain signals. However, the DRG contains more than just nerve cells, and we were surprised to find that the differences between acute and chronic neck pain were not in the nerve cells themselves.
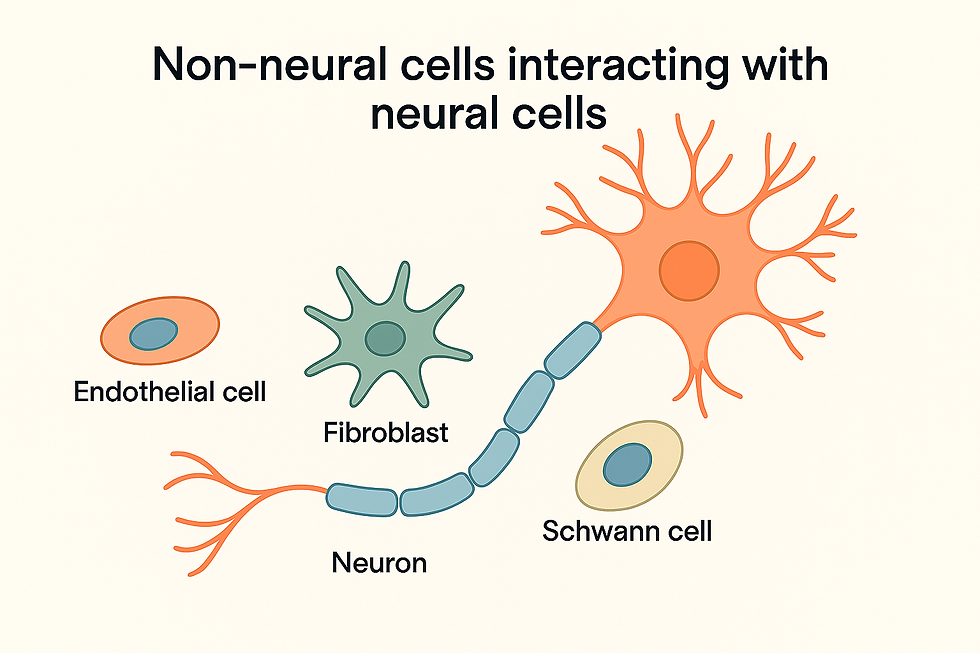
Instead, the major changes were seen in cells that surround and interact with nerve cells, including:
Cells lining blood vessels (endothelial cells)
Cells that help organize and support tissue (fibroblasts)
Cells that insulate nerve fibers (Schwann cells)
These cells have several functions, including regulating the passage of substances from the blood into the tissues, supporting nerve health, and promoting nerve repair.
Ongoing inflammation around the nerves
In people with chronic neck pain, these non-neural cells showed strong signs of ongoing inflammation. Many of the more active genes are known to be involved in inflammation, immune signaling, and pain sensitivity. This suggests that non-neural cells may send chemical signals to nearby nerve cells, keeping them in a pain‑sensitive state. Consequently, chronic neck pain may be driven by an "unhealthy" inflammatory environment around the nerves.
At the same time, we also saw increased activity in genes that may help protect nerves or dampen pain. This suggests that the body may be trying to reduce the pain.
Why this matters for patients
This is the first molecular study of the human C2 DRG at a single-cell resolution. By studying human tissue from patients with pain and linking it to patients’ pain experiences, we are laying the groundwork for the development of effective treatments for chronic neck pain. Our hope is that this research will help move the field toward therapies that address the biological drivers of persistent neck pain and ultimately improve patients' quality of life.
Acknoledgment
This research has been made possible by funding from the National Institute of Health (NIH).
Reference
[1] Arendt-Tranholm A, Sankaranarayanan I, Payne C, Mancilla Moreno M, Mazhar K, Yap N, Chiu AP, Barry A, Patel PJ, Inturi NN, Tavares-Ferreira D, Amin A, Karandikar M, Jarvik JG, Turner JA, Hofstetter CP, Curatolo M, Price TJ. Transcriptome of the human C2 dorsal root ganglia in C1-2 arthrodesis surgery: insight for neck pain. Brain. 2026 Apr 7;149(4):1349-1364. doi: 10.1093/brain/awaf370. PMID: 41032656; PMCID: PMC13058466.


